
The world’s Largest Sharp Brain Virtual Experts Marketplace Just a click Away

Levels Tought:
Elementary,Middle School,High School,College,University,PHD
| Teaching Since: | May 2017 |
| Last Sign in: | 408 Weeks Ago, 5 Days Ago |
| Questions Answered: | 66690 |
| Tutorials Posted: | 66688 |
MCS,PHD
Argosy University/ Phoniex University/
Nov-2005 - Oct-2011
Professor
Phoniex University
Oct-2001 - Nov-2016
Question 14
During the kinetic study of the reaction, 2A + B ? C + D, following results were obtained:
|
Run |
[A]/mol L–1 |
[B]/mol L–1 |
Initial rate of formation of D/mol L–1 min–1 |
|
I |
0.1 |
0.1 |
6.0 × 10–3 |
|
II |
0.3 |
0.2 |
7.2 × 10–2 |
|
III |
0.3 |
0.4 |
2.88 × 10–1 |
|
IV |
0.4 |
0.1 |
2.40 × 10–2 |
Based on the above data which one of the following is correct? [CBSE AIPMT 2010]
| A |
rate = k[A]2 [B]2 |
| B |
rate = k[A] [B]2 |
| C |
rate = k[A]2 [B] |
| D |
rate = k[A] [B] |
| Question 15 |
For the reaction N2O5(g) ? 2NO2(g) + 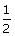 O2(g) the value of rate of disappearance of N2O5 is given as 6.25 × 10–3 mol L–1 s–1. The rate of formation of NO2 and O2 is given respectively as: [CBSE AIPMT 2010]
O2(g) the value of rate of disappearance of N2O5 is given as 6.25 × 10–3 mol L–1 s–1. The rate of formation of NO2 and O2 is given respectively as: [CBSE AIPMT 2010]
A
6.25 × 10–3 mol L–1 s–1 and 3.125 × 10–3 mol L–1 s–1
B
6.25 × 10–3 mol L–1 s–1 and 6.25 × 10–3 mol L–1 s–1
C
6.25 × 10–3 mol L–1 s–1 and 6.25 × 10–3 mol L–1 s–1
D
1.25 × 10–2 mol L–1 s–1 and 3.125 × 10–3 mol L–1 s–1

Hel-----------lo -----------Sir-----------/Ma-----------dam-----------Tha-----------nk -----------You----------- fo-----------r u-----------sin-----------g o-----------ur -----------web-----------sit-----------e a-----------nd -----------and----------- ac-----------qui-----------sit-----------ion----------- of----------- my----------- po-----------ste-----------d s-----------olu-----------tio-----------n.P-----------lea-----------se -----------pin-----------g m-----------e o-----------n c-----------hat----------- I -----------am -----------onl-----------ine----------- or----------- in-----------box----------- me----------- a -----------mes-----------sag-----------e I----------- wi-----------ll