
The world’s Largest Sharp Brain Virtual Experts Marketplace Just a click Away

Levels Tought:
Elementary,Middle School,High School,College,University,PHD
| Teaching Since: | May 2017 |
| Last Sign in: | 408 Weeks Ago, 3 Days Ago |
| Questions Answered: | 66690 |
| Tutorials Posted: | 66688 |
MCS,PHD
Argosy University/ Phoniex University/
Nov-2005 - Oct-2011
Professor
Phoniex University
Oct-2001 - Nov-2016
1. A spring-loaded piston/cylinder contains 1.5 kg air at 27â—¦C, 160 kPa. It is now heated in a process in which pressure is linear in volume, P = A + BV, to twice its initial volume, where it reaches 900 K. Find the work, the heat transfer, and the total entropy generation, assuming a source at 900 K.
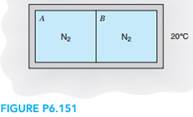
2. A rigid container with volume 200 L is divided into two equal volumes by a partition, shown in Fig. P6.151. Both sides contain nitrogen; one side is at 2 MPa, 200â—¦C and the other at 200 kPa, 100â—¦C. The partition ruptures, and the nitrogen comes to a uniform state at 70â—¦C. Assume the temperature of the surroundings is 20â—¦C. Determine the work done and the net entropy change for the process.

Hel-----------lo -----------Sir-----------/Ma-----------dam-----------Tha-----------nk -----------You----------- fo-----------r u-----------sin-----------g o-----------ur -----------web-----------sit-----------e a-----------nd -----------acq-----------uis-----------iti-----------on -----------of -----------my -----------pos-----------ted----------- so-----------lut-----------ion-----------.Pl-----------eas-----------e p-----------ing----------- me----------- on-----------cha-----------t I----------- am----------- on-----------lin-----------e o-----------r i-----------nbo-----------x m-----------e a----------- me-----------ssa-----------ge -----------I w-----------ill----------- be-----------